 
This occurs because of “a trade-off between correlations and entropy,” said Roberto Serra, a physicist at the Federal University of ABC and the head of the research group behind the study. 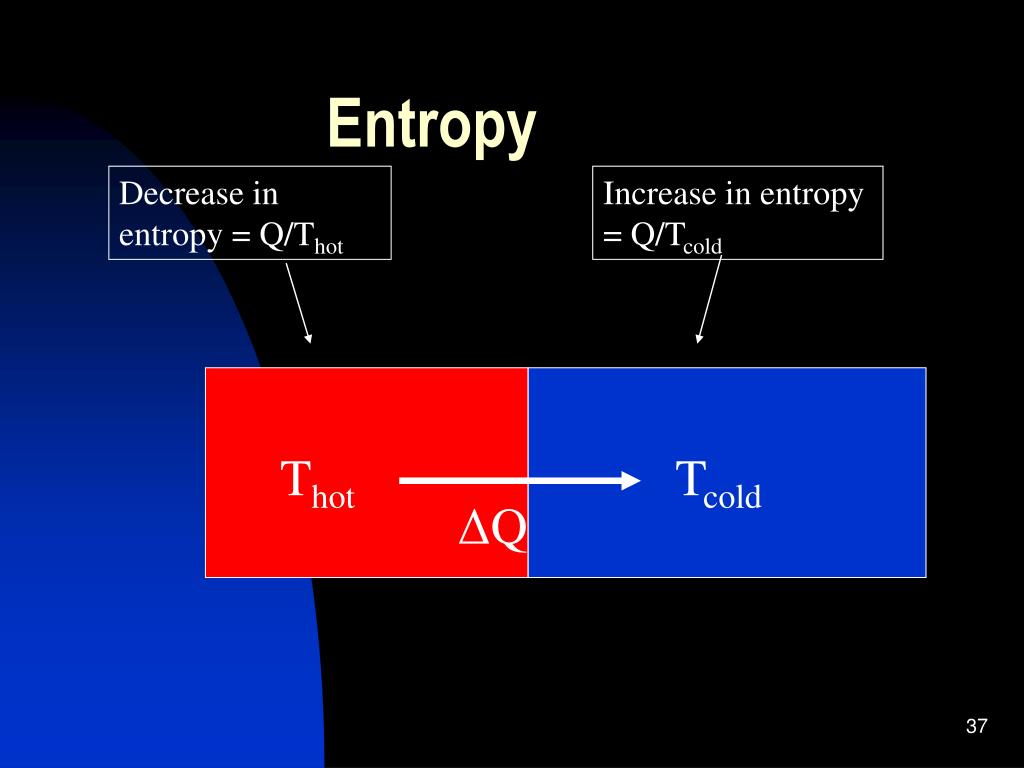
In other words, heat flows from cold to hot. The cold qubit gets colder, the hot qubit hotter. The weakening of the correlation is akin to a “fuel driving the heat from the colder to the hotter body,” said David Jennings, a physicist at Imperial College London. But here, the researchers take the correlation into account. If the total entropy suddenly decreased in a regular, uncorrelated system, it would violate the second law. 
“That means that the sum of the individual entropies decreases,” said Kaonan Micadei, a researcher at the Federal University of ABC in São Paulo, Brazil, who participated in the study. As the experiment progresses, the particles gradually unlink themselves, and the correlation weakens. The experiment begins with the two particles strongly correlated. By comparing the entropy of the single qubits to the entropy of the correlated system, researchers can measure the amount of correlation. A two-qubit system can be in one of four possible states - denoted by 00, 01, 10 and 11 - and its entropy is defined by the probability of being in each of these states. In the quantum world, correlations affect entropy. In a classical system, the entropy of a system is equal to the sum of the entropies of each of its parts. Traditionally, entropy is the measure of the number of different configurations a system can be in. These correlations made the puzzling behavior possible. This caused the nuclei to become linked, or correlated, turning them into a single, inseparable whole, a two-qubit quantum state. They then generated a magnetic field to align the nuclear spins of the two quantum particles, or “qubits” - the carbon and hydrogen nuclei. The team, based in Brazil, took a molecule that consisted of a carbon atom, a hydrogen atom and three chlorine atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
